Empowering the natural killers in our immune system
What if we could activate and empower the natural killers in our immune system to target leukaemia cells? Could we remove the debilitating side-effects of less-targeted treatments and transform people’s lives?
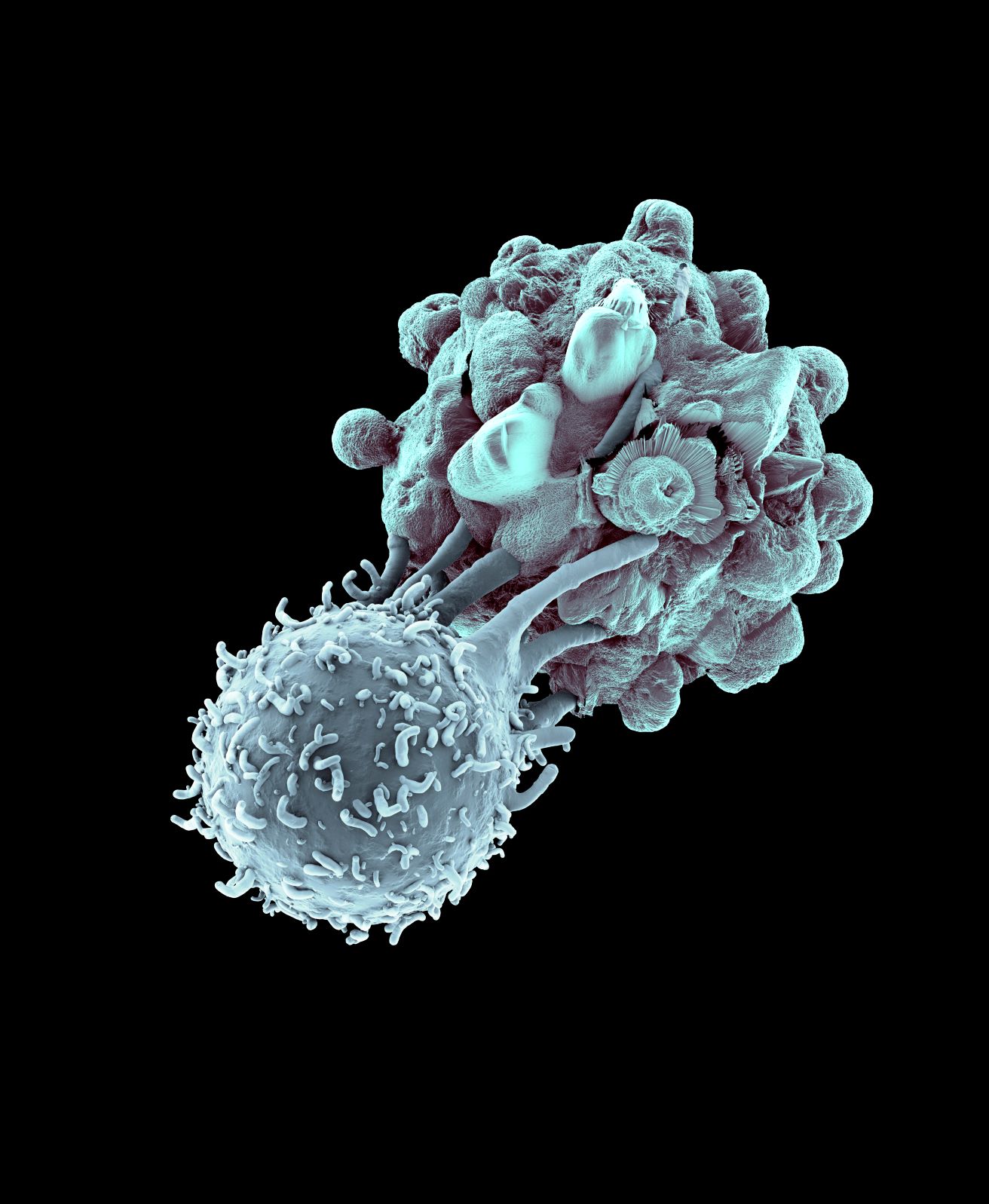
Dr Matt Blunt, Leukaemia UK John Goldman Fellow, has found new ways to direct one group of immune cells against leukaemia – discoveries which could lead to effective treatments with fewer side-effects to save more lives.
 People with leukaemia often face difficult treatment options. But a new class of treatments has emerged over the last decade which promise both effectiveness and fewer side-effects: immunotherapies. The immune system is seen as the body’s ultimate line of defence, and the prospect of harnessing it for treating cancer is very attractive.
People with leukaemia often face difficult treatment options. But a new class of treatments has emerged over the last decade which promise both effectiveness and fewer side-effects: immunotherapies. The immune system is seen as the body’s ultimate line of defence, and the prospect of harnessing it for treating cancer is very attractive.
Among the various immune cells which could be used against cancer, T cells have received the biggest focus, thanks to the excitement around CAR-T cell therapies. But researchers are now looking to other immune cells in the search for new and effective treatments for leukaemia. One such scientist is Dr Matt Blunt, who studies immune cells called Natural Killer (NK) cells.
“A lot of the focus has been on T cells, and I think partly that’s been because NK cells are maybe a little bit harder to detect”, says Matt. He explains that this is partly because there’s just a lot more T cells than NK cells.
Finding out what makes a natural killer
Working at the University of Southampton, Matt has been studying how NK cells might be recruited to attack B-cell cancers, including chronic lymphocytic leukaemia (CLL) and acute lymphoblastic leukaemia (ALL), but also many common lymphomas.
At first, Matt was particularly interested in NK cells which carry a molecule on their surface called KIR2SD2. Roughly half of the population carry this molecule on their NK cells. Previous research has shown that people with blood cancer who receive a stem cell transplant from donors who have KIR2DS2 have a lower chance of their cancer getting worse or coming back.
However, it was not clear why this is the case, and what exactly KIR2DS2 is doing to these NK cells. So Matt was awarded the John Goldman fellowship from Leukaemia UK [hyperlink] in 2019 to find out.
In his research, Matt confirmed that NK cells which carry KIR2DS2 are primed for killing cancer cells, and his experiments revealed why. As well as KIR2DS2, these cells have high levels of a molecule called CD16 on their surface, which is a key player in how NK cells kill cancer cells. Matt showed this means these KIR2DS2-positive cells are naturally more active against all kinds of cancers, including ALL and CLL, but also lymphomas and even liver cancers.
Could this be Dr Blunt’s most important discovery?
Matt suspected that activating these KIR2DS2-positive NK cells may help make treatment against leukaemias and other blood cancers more effective. So, during his fellowship, Matt developed a new way to activate NK cells, using a peptide – a small fragment of a larger protein, in this case a protein called exportin-1.
It was this that led to a surprising discovery that might be one of Matt’s most important yet.
Using natural killer cells to boost cancer drugs
Exportin-1 is a target for a drug called selinexor, an exciting treatment which kills cancer cells in a completely unique way. One of selinexor’s effects is that it causes exportin-1 to be broken up into peptides – the same kind of peptides that could activate the NK cells Matt was studying.
So, Matt wanted to see if combining selinexor with the KIR2DS2-positive NK cells might make these special NK cells even more effective against blood cancer.
However, Matt’s experiments showed that all NK cells were activated by selinexor, whether they had KIR2DS2 or not. Further research revealed that selinexor was activating NK cells in a completely different way to how he expected.
“It was a complete surprise to us, actually,” Matt says. For him and his colleagues, using selinexor “started off as a tool for us to study KIR2DS2. But then it just led in this completely unexpected path that’s now really taken off.”
Kinder and more effective immunotherapies
Selinexor is in trials for a range of different cancer types and is currently approved for treating multiple myeloma. To make it useful for treating leukaemia, Matt is working with the pharmaceutical company that developed selinexor, looking to combine it with other drugs already approved for leukaemia, or with new NK cell therapies.
Right now, Matt and his team are testing these ideas in the lab and are talking to clinicians about clinical trials in the not-too-distant future. “We could set up a trial in patients with leukaemia and give them selinexor plus an NK therapy,” Matt explains.
“I think that would be really fantastic, and that’s what we’re in discussions of at the moment. I’m really hoping that will take off.”
Therapies benefitting more people across the world
To explain why Matt is so excited about the potential of NK cell therapies for leukaemia, it helps to compare them to CAR-T cell therapies. Because of the way that NK cells work, they could cause fewer severe side-effects than CAR-T cell therapies. Another benefit of NK cells is that can be donated by any donor and given to any patient, meaning they could become an ‘off the shelf’ treatment, instead of the tailor-made but laborious process of creating CAR-T cells. This would make NK cell therapies quicker and cheaper to administer, meaning more people across the world could benefit.
Matt’s work adds to the growing evidence that NK cells have more of a part to play in cancer treatment than previously thought. “I think NK cells may have been under-appreciated,” he says, “but it’s becoming increasingly clear that they have an important role.”
The difference that Leukaemia UK funding made
Being awarded a John Goldman Fellowship from Leukaemia UK has not only enabled Matt to study NK cells as leukaemia treatments. It has also helped him make an important step in his career: from a researcher in someone else’s lab group, to setting up his own team and his own programme of research.
“I think that’s a really key stage of people’s careers,” he says. “There isn’t that much funding available like this for early career researchers.”
Funding like the John Goldman Fellowships gives more scientists the opportunity to put their unique research experience into practise. Matt believes this benefits the field of leukaemia research and, in turn, people with leukaemia. “People can come in with new ideas and do new research that wouldn’t have been taking place before.”
Support our life-saving research by donating to Leukaemia UK.
